Mitocholine® is novel composition of fixed ratio of three human essential human metabolites: Choline, Succinate and Nicotinamide, which, when taken orally, become available to the brain cells.
Mitocholine® is our breakthrough solution for fuelling the brain and supporting cognitive performance. It is uniquely formulated as functional nutritional product Disuccinate salt of Choline with addition of Nicotinamide. Mitocholine® addresses the high energy needs of the brain because it has a synergistic effect on both ATP and NADH generation and on phosphocreatine (pCr) level in the brain.
Mitocholine® enhances brain energy stores for optimal health and functioning.
Although Mitocholine® represents a completely unique, patented and novel combination of the above bioactive ingredients there is a long history of human exposure to its key components.
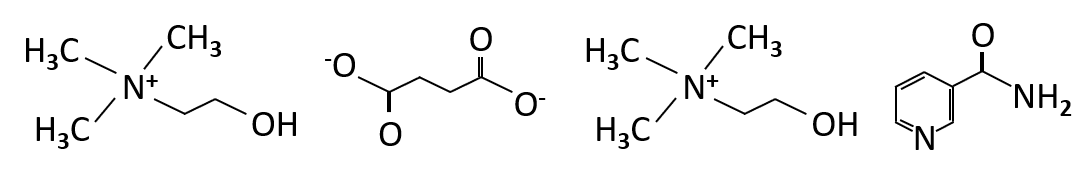
Choline
Humans can produce small amounts of choline, however, most individuals must consume this nutrient through the diet to supplement the endogenously produced amount to prevent deficiency. In the body choline is a component of the key brain neurotransmitter acetylcholine as well as other brain and nervous tissues. Choline is also a dietary element that is vital for the structural integrity of cell membranes, transmembrane signalling, lipid and cholesterol transport, methyl metabolism. Choline availability modulates brain development in the foetus and infant, and in the adult is important for normal liver and muscle function (1). Loss of cholinergic neurones is associated with impaired cognitive function, memory loss and Alzheimer’s disease (2).
In the EU there are three authorised health claims for choline.
Choline contributes to normal homocysteine metabolism. (3)
Choline contributes to normal lipid metabolism. (3)
Choline contributes to the maintenance of normal liver function. (3)
Learn More
According to US consumption data from 2007-2010, 92% of the US population failed to achieve daily recommended adequate intake (AI) of choline (4, 5).
The European Food Safety Agency (EFSA) has set an AI of from 140 mg/day to 520 mg/day depending on population group (6).
Human clinical trials demonstrated link between dietary choline intake and cognitive function.
References:
1. https://www.ncbi.nlm.nih.gov/pubmed/31385730
2. https://pubmed.ncbi.nlm.nih.gov/25290336
3. https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX:32012R0432
4. https://www.ers.usda.gov/data-products/food-consumption-and-nutrient-intakes
5. https://www.ncbi.nlm.nih.gov/pubmed/26886842
6. https://efsa.onlinelibrary.wiley.com/doi/epdf/10.2903/j.efsa.2016.4484
Succinate
Succinate is formed endogenously as a component of the Tricarboxylic acid cycle (TCA), the final common oxidative pathway for carbohydrates, fats and amino acids. The Scientific Committee on Foods (SCF) concluded for succinic acid in 1990.
“The anion occurs in nature and plays a role as an intermediate metabolite in the citric acid cycle”.
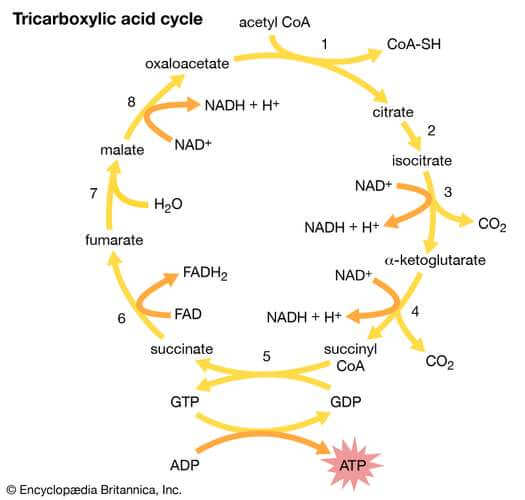
Succinic acid is therefore present in almost all plant and animal tissues. Succinic acid is approved for use in foods as a flavour enhancer and pH control agent (1).
In the US, succinic acid is permitted for addition to food as an ingredient Generally Recognised as Safe (GRAS) (2).
Succinate, is substrate for the cellular energy making machinery in mitochondria as well as a signalling molecule reflecting the cellular metabolic state (3).
Learn More
In 2007 the mechanism of the mitochondrial control of insulin receptor activation in the brain has been discovered. It was demonstrated that mitochondrial substrate -succinate facilitates neuronal insulin receptor activation. Succinate in the form of highly bioavailable salt of choline promotes and potentiates intracerebral insulin functions in the brain. These functions include effects on concentration, memory and mood and are mediated through activation of neuronal isoform of the insulin receptor (4, 5, 6). Succinate was also identified as specific substrate that supports the TCA cycle and ATP generation by oxidative phosphorylation in stressed cells when mitochondrial function is impaired (7).
References:
1. http://www.foodchemadditives.com/applications-uses/1627
2. http://www.accessdata.fda.gov
3. https://pubmed.ncbi.nlm.nih.gov/26971832
4. https://pubmed.ncbi.nlm.nih.gov/18215309
5. https://pubmed.ncbi.nlm.nih.gov/23730255
6. https://pubmed.ncbi.nlm.nih.gov/24094269
7. https://pubmed.ncbi.nlm.nih.gov/20566649
Nicotinamide
Nicotinamide (NAM) is a soluble organic compound belonging to the group of B vitamins. Along with nicotinic acid, nicotinamide is often referred to by the generic name niacin which is found in a wide range of foods. In the EU there are six authorised choline health claims for niacin:
Niacin contributes to normal psychological function (1).
Niacin contributes to normal energy-yielding metabolism (1).
Niacin contributes to normal functioning of the nervous system (1)
Niacin contributes to the maintenance of normal mucous membranes (1).
Niacin contributes to the maintenance of normal skin (1).
Niacin contributes to the reduction of tiredness and fatigue (1).
1. https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX:32012R0432
Learn More
Nicotinamide (NAM), also known as niacinamide and niacin is a form of vitamin B3 found in food and used as medication and supplement (2, 3).
The main sources of niacin include liver, meat (or meat products), fish, peanuts and whole grains. Foods rich in protein, such as milk, cheese, and eggs, which are good sources of the amino acid tryptophan, are therefore good sources of niacin equivalents (NEs). Niacin can be synthesized in the human body from the amino acid tryptophan. However, this synthesis is dependent on the levels of other vitamins and minerals in the body and low levels of iron, riboflavin or vitamin B6 decrease the conversion of tryptophan to niacin (4).
Nicotinamide is an essential precursor to the NAD+ coenzyme used for turning nutrients into energy. NAD+ transfers electrons in redox reactions to help synthesize ATP, the energy currency of the cell. NAD+ molecule is critical in regulating cellular aging and is a substrate for mitochondria. NAD+ is the most researched and cited molecule in anti-ageing research at present. NAD+ decreases over time and in metabolic stress. Increase in NAD+ maintains healthy mitochondria, promotes cellular repair and maintains steady healthy levels of energy production and cellular metabolism (5, 6).
Recently NAD+ has been proven as an important signalling molecule required for th activity of sirtuins (SIRTs), PARPS polymerases and cyclic ADP-ribose synthases. NAD+ continious turnover is tightly linked to metabolic homeostasis (7).
Every pair of electrons donated by the NADH in the electron transport chain form 2.5 ATP molecules. It is approximately 80 kg of ATP turned over processed by a male adult each day, it is predicted as much as 30 kg NADH is generated every day in the human body. In mammalian cells there are few different pathways have been established from Nicotinamide, nicotinic acid and nicotinamide riboside. NAM and and NR contain an amine group and follow an amidated route to NAD+ (8, 9).
History:
NAD+ has long been associated with disease with symptoms of pellagra first described in the 1800, caused by malnutrition or by deficiency in vitamin B3, pellagra is characterised by three symptoms dermatitis, diarrhoea and DEMENTIA. Supplementation with NAD+ precursors successfully treated pellagra (10).
Modern anti-aging research is aimed at defining the key pathways to sustain high NAD+ turnover. The notion of using NAD+ precursors to boost NAD+ is now beginning to be translated into human studies, NAD+ precursors are being safe and naturally available and having potential to improve human health and longevity.
Click to see
References:
2. https://pubmed.ncbi.nlm.nih.gov/22646128
3. https://pubmed.ncbi.nlm.nih.gov/18429699
4. https://pubmed.ncbi.nlm.nih.gov/17161604
5. https://pubmed.ncbi.nlm.nih.gov/18335035
6. https://pubmed.ncbi.nlm.nih.gov/21589930
7. https://pubmed.ncbi.nlm.nih.gov/29697773
8. https://pubmed.ncbi.nlm.nih.gov/25423279
9. https://pubmed.ncbi.nlm.nih.gov/31412683
10. https://pubmed.ncbi.nlm.nih.gov/16207585
11. https://pubmed.ncbi.nlm.nih.gov/29249689

MitocholineTM - maintaining brain health through nutrition
